4D MRI to measure fetal blood flow
The fetus is remarkable in that it grows and develops in an environment of physiological hypoxaemia and hypoglycaemia relative to the mother.
Read MoreThe fetus is remarkable in that it grows and develops in an environment of physiological hypoxaemia and hypoglycaemia relative to the mother.
Read More
One of the leading causes of blindness in the Western world are vascular diseases affecting the retina. The arteries and veins inside the light-sensitive layer in the back of the eye – the retinal vasculature – are an intricate network supplying the inner retina with crucial nutrients while removing metabolic waste. The heterogeneity in space and time of blood flow in this microvasculature is critical as the retina has one of the highest metabolic demands of the central nervous system.
Pathological retinal neovascularisation and oedema – that is swelling, thickening, or unusual growth in the retinal vasculature – are commonly responsible for certified visual loss. Therefore, understanding the spatial and temporal heterogeneity and active regulation of retinal blood flow in the retina is critical for the diagnosis of retinal vascular disease. Furthermore, these blood vessels are affected by systemic vascular diseases and, as such, evidence of these conditions may be observed through the microvasculature inside the eye.
Recently, a technique known as optical coherence tomography angiography (OCTA) has been developed as a non-invasive approach to visualising blood vessels. This method uses the reflectance of light on the surface of moving blood vessels to map the vasculature of the retina.
A multi-disciplinary team, headed by Prof Dao-Yi Yu at the Lions Eye Institute (affiliated with the UWA Centre for Ophthalmology and Visual Science) are using OCTA to reveal remarkable spatial and temporal heterogeneity of retinal capillary perfusion. The project aims to use OCTA as a non-invasive tool for the early detection of retinal vascular diseases.
NIF Informatics Fellow, Dr Andrew Mehnert, is contributing his analysis expertise to guide improvements in OCTA instrumentation and algorithms. Image analyses, undertaken in the Characterisation Virtual Laboratory (CVL) using both MATLAB and FIJI/ImageJ, showed remarkable resolution of capillary perfusion in the macular region of human and porcine subjects.
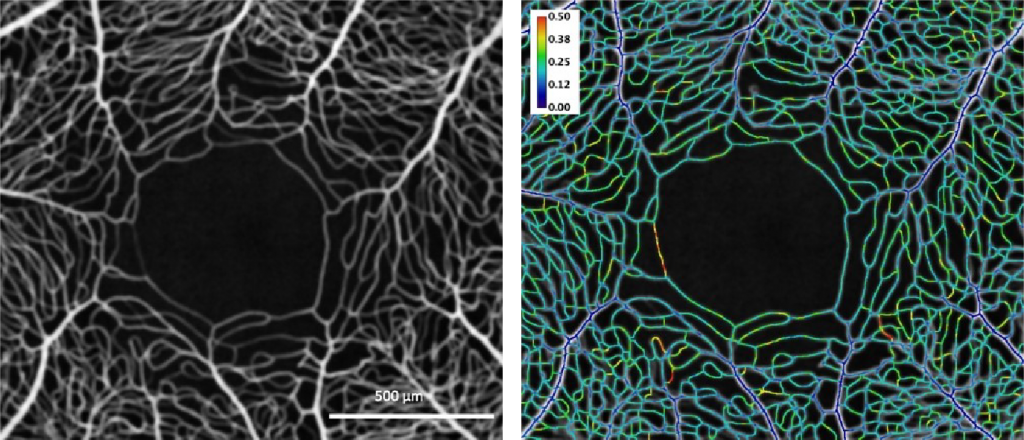
Left: En face mean intensity projection through 31 OCTA images from the macular region of a human subject. Right: Coefficient of variation along vessel centrelines showing spatial and temporal heterogeneity of capillary perfusion. The colour bar indicates the variation from dark blue (no variation) through to red (most variation).
The developed software tools have been used for characterising spatio-temporal variation of capillary perfusion in OCTA images.
Experimental results to date are both valuable and encouraging because they may be potentially useful for clinical diagnostic purposes and can be used to guide improvements in OCTA instruments and new algorithm development. These results move towards a non-invasive tool for the early detection of retinal vascular diseases in humans, and may also be used for other investigations of capillary perfusion where the retina is an appropriate model for microcirculation studies.
For further information, please contact Dr Andrew Mehnert.
This story was contributed by the University of Western Australia.
Over 250,000 Australians currently live with epilepsy, a chronic disorder of the brain. In addition to significant social, physical and psychological consequences, epilepsy is linked with an increased risk of death compared to the general population. Up to 70% of epilepsy cases may be controlled through lifestyle and medication; however, many patients are unable to be treated using conservative measures. A major clinical problem, drug-refractory focal epilepsy may require aggressive treatments, including surgery, to resect a portion of the brain.
The localisation of the epileptogenic zone (the portion of the brain requiring resection to reduce seizures) is typically carried out clinically using magnetic resonance imaging (MRI). In many cases, no abnormalities are detected, requiring further investigation. Surgeons may also use invasive surgically implanted electrodes (intracranial electrode depth or grid, or ICEEG) for seizure source localisation, increasing the risk to the patient. A lower risk option is further functional imaging with positron emission tomography (PET), single-photon emission computed tomography (SPECT), and simultaneous electroencephalography functional MRI (EEG-fMRI). Unfortunately, many of these modalities lack temporal resolution to distinguish seizure onset.
“Catching the earliest wave, the earliest is key”
Representation of MEG (green) and EEG (blue) overlays.
Magnetoencephalography (MEG) and electroencephalography (EEG) offer a non-invasive method to detect rapidly evolving brain wave spikes and seizures at millisecond temporal resolution. A/Prof Chris Plummer and team, Mr Simon J. Vogrin, Dr William P. Woods, A/Prof Michael A. Murphy, Prof Mark J. Cook, and Prof David T.J. Liley, have developed a world-class multimodal technique for non-invasive source localisation and epileptogenic zone characterisation. The method (using co-acquired MEG and high-density EEG) has already improved the surgical treatment of medically refractory epilepsy, with clinical validity demonstrated in a key 2019 publication.
The team utilised the world-class MEG at the SUT NIF Node together with the expertise of NIF Facility Fellow Dr Will Woods to look at the efficacy of concurrent HDEEG (High-Density EEG > 64 electrodes) and MEG for identifying the epileptogenic zone. Ninety-six patients have been involved in the long-term prospective study. Seventy-one patients were MRI negative and twenty-five MRI positive with either complex lesions or multiple lesions. More than five years’ data collection in collaboration with neurologists and medical scientists found that the combined method was able to detect interictal spike discharges (between seizures) at early, mid and late-phase as well as ictal discharges (during seizures). The earliest resolvable localisation by either EEG or MEG was determined to be the most suitable site for surgical resection.
“We find that for most cases, modelling either the mid-upswing or the later peak (still common practice) results in mislocalization of the cortical generator (and the putative Epileptogenic Zone) due to the effects of discharge propagation across the cortical surface. We look for the earliest feature from the discharge; sometimes it appears first in the Magnetometer, sometimes it is first in one of the Gradiometers, and sometimes it is first in the HDEEG. For a given patient, there is typically a dominant leading modality, but the modality that leads in time will vary from patient to patient. This is fundamentally the reason for recording both. Otherwise, you risk having half the story before diving into invasive investigations.” – A/Prof Plummer
To date, successful source localisation has guided successful surgical outcomes in 25 patients, 8 of whom were spared invasive intracranial monitoring due to this method. Many of the over 100 studies performed are yet to progress to surgery, underpinning the latent potential of yet to be followed complete surgical workup and seizure-freedom outcomes following surgical therapy.
“Accurate, non-invasive localisation with this method will reduce the medical burden of invasive intracranial electrode monitoring by reducing the number of invasive electrodes required and, in some cases, remove the need for these in the first place to allow the patient to proceed directly to potentially curative surgery.” – A/Prof Plummer
This work intends to revolutionise epilepsy management in Australia, with data from a larger and more diverse cohort expected to provide evidence for a Medicare rebate to facilitate the accessibility of this technique for Australians.
Success stories:
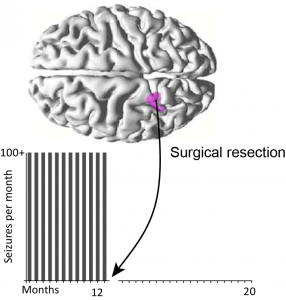
From 500 seizures per month to none; MEG/EEG guided surgical resection success
A young man with a five-year history of disabling seizures was suffering up to 500 seizures per month; his schooling was disrupted, and all the standard non-invasive tests gave conflicting results. Surgical resection was performed correlating with the earliest EMSL result. The patient is now seizure-free and on minimal medication following surgery.
A 10-year old child had a four-year history of left leg motor-sensory events, up to 200 times per day, leading to falls. PET and MRI suggested localisation of seizures in the right superior frontal gyrus and the right paracentral lobule, while EMSL results showed seizure onset further to the rear at the precuneus. Surgical resection of the paracentral lobule and superior frontal gyrus did not relieve the seizures seen by ICEEG during surgery. Further exploration revealed a second pocket of dysplasia in line with the EMSL result, resection of which normalised the ICEEG and relieved the patient of seizures.
A 36-year-old patient presented with speech-arresting complex partial turns with a typical MRI result. Their surgical plan was altered to include a hippocampal depth electrode, where seizures were found to start by MEG. Standard anteromesial temporal lobectomy revealed a cortical dysplasia involving the entorhinal cortex. The patient is now seizure-free and works as a speech therapist.
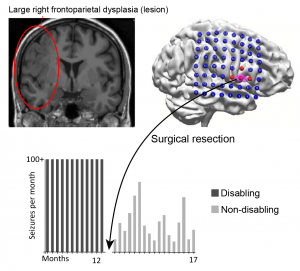
Recovering day to day activities without disturbing the complex dysplasia
A 36-year old mother of two young children suffered a 33-year history of multiple-daily disabling motor seizures. She was denied surgery due to a complex lesion seen by MRI (frontoparietal dysplasia). EMSL captured the seizure onset, targeting surgical resection without interfering with the bulk of the lesion. The patient is now on reduced medication and experiences only brief non-disabling focal seizures. She states that she “has her life back”.
You can learn more about epilepsy here or by speaking to your GP. This work was contributed by St Vincent’s Hospital, Swinburne University of Technology, and the University of Melbourne. For more information, please contact Dr Will Woods.
The lymphatic system is one of the key mechanisms for metastatic spread, whereby cancer cells that have disseminated from a primary tumour are taken up into lymphatic vessels and transported to other locations in the body, beginning with the lymph nodes. Conventionally, surgeons map the lymphatic flow from a tumour site into the first draining nodes – known as sentinel lymph nodes – using a Tc99m radiocolloid. Drainage pathways of tracers may be imaged before surgery with gamma cameras (lymphoscintigraphy), and individual nodes detected during surgery with handheld gamma probes. These sentinel nodes are the ‘canary in the coal mine’ and can offer valuable information to help stage the disease and determine the most appropriate treatment options, so it is ideal for removing just these nodes while leaving uninvolved nodes intact.
While this technique is successfully and routinely applied to breast cancer and melanoma, accuracy is limited in cancers where the nodes are in close proximity to the primary tumour or other nodes, such as cancers of the head and neck. Without an accurate method to identify sentinel nodes in oral cancer, extensive dissection of all nodes in the neck region is routinely performed to ensure any potential draining nodes are harvested, yet approximately 75 % of patients undergoing neck dissection are exposed to the complications and morbidity of this invasive procedure without clinical benefit.
Dr Aidan Cousins and Dr Giri Krishnan have been working as part of a collaborative project between the University of South Australia and the University of Adelaide to demonstrate a novel, high-resolution technique for lymphatic mapping using magnetic nanoparticle-based tracers. Studies conducted at the South Australian Health and Medical Research Institute (SAHMRI) involve the injection of the magnetic tracer to the oral cavity of female Large White pigs, which are then scanned with a 3.0T Siemens MRI located at the large animal research and imaging facility (LARIF) NIF Node. Post-injection scans give Dr Cousins and Dr Krishnan detailed anatomical information of the drainage patterns of the tracer, which is then used to plan the surgery. During surgery, a handheld magnetometer probe developed by Dr Cousins is used, along with MRI data and visual identification, to pinpoint the draining nodes of interest from other, uninvolved nodes.
Dr Cousins with Raj Perumal (LARIF) examining an MRI scan of a pig following injection of the magnetic tracer
The results of this experiment showed the magnetic alternative to be adept for mapping lymphatic drainage in complex environments. Triangulating the location draining nodes with high precision before surgery was made possible by the high-resolution soft-tissue detail afforded by the 3.0T MRI. During surgery, the handheld probe was able to identify all draining nodes by way of detecting their magnetic ‘signature’. This final confirmation of draining nodes is analogous to the use of handheld gamma probes but has the distinct advantage of pin-point resolution, meaning nodes can be in very close (touching) proximity to each other and it is still possible to differentiate between the individual nodes’ magnetic signals. This spatial resolution is currently unmatched by any other probe technology commercially available and is key to the application of sentinel node mapping in complex cancers.

Photo of the high-resolution magnetometer probe developed by Dr Cousins
Results from this study are being used to design a world-first clinical trial applying magnetic tracers and a high-resolution probe to human patients with cancer of the oral cavity.
This story was contributed by SAHMRI. For further inquiries, please contact Mr Raj Perumal
The arches of the human foot are unique structures that are important for functions like walking and running. The deep intrinsic muscles of the foot, such as the adductor halluces and interossei, are thought to play key roles in arch control; yet little is known about how they are controlled during functional tasks. The traditional measurement techniques can only provide information regarding muscle size, which is inadequate to evaluate the force-generating capacity of the muscles, assess the process of force generation by the muscles, and understand the involvement of the neural drive sent by the body to the muscles to regulate force production.
Using the 7T human MRI at the Centre for Advanced Imaging, a research study is currently investigating the muscle architecture of the adductor halluces and interossei. This research aims to quantify the force-generating capacities of these deep-foot muscles by measuring their MRI muscle volumes, estimate their force production by shear wave Elastography and measure their neural drive by using Electromyography.
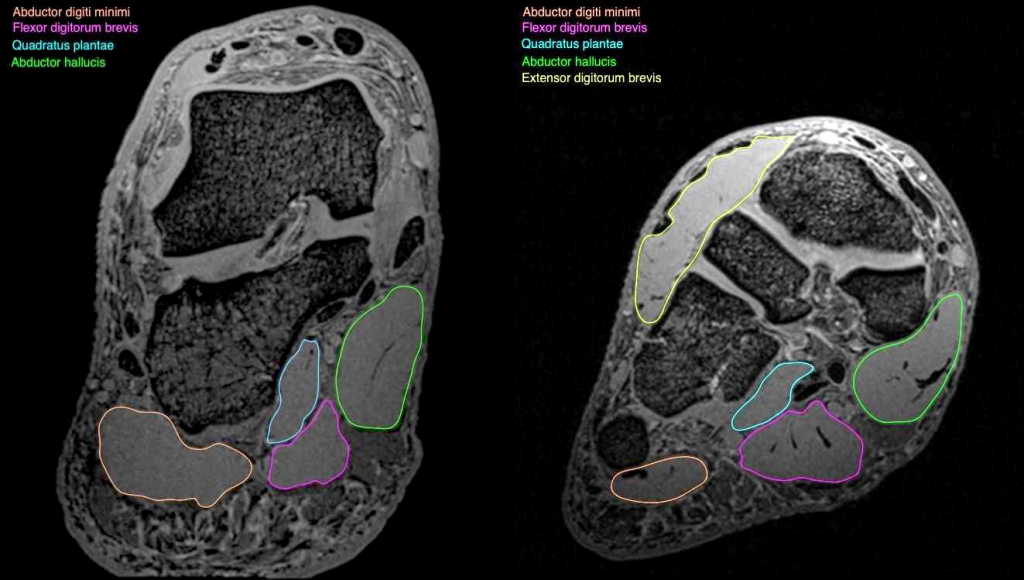
Deep foot muscles outlined in two cross-sections of a structural MRI scan obtained at ultra-high field: abductor halluces (green), abductor digiti minimi (pink), flexor digitorium brevis (magenta), quadrature plantae (cyan) and extensor digitorium brevis (yellow). With thanks to Dr Natalie Collins, University of Queensland
MRI images are obtained from male and female volunteers with no lower limb pain or injury. To date, structural MR images of deep foot muscles have been obtained using T1 VIBE 3D Transverse Oblique sequence and MRI muscle volumes have been measured. In future, diffusion tensor imaging will be performed to measure apparent diffusion coefficient and fractional anisotropy of the deep foot muscles.
This story was contributed by the University of Queensland, with acknowedgements to Dr Natalie Collins of the School of Health and Rehabilitation Sciences, Health and Behavioural Sciences, University of Queensland.
For further information, please contact Dr Tonima Ali.
Diffusion tensor imaging (DTI) is a magnetic resonance imaging (MRI) technique that exploits the movement of water molecules to reveal microscopic details about tissue architecture. DTI is commonly used in brain imaging studies, used to track neural tracts through the brain. The technique is also ideal for investigating the 3D architecture of muscles, as DTI can be used to obtain detailed, quantitative measurements of the anatomy of complex skeletal muscles in living humans. Prof Robert Herbert’s group at NeuRA utilised the 3T MRI located in the UNSW Node of NIF to take a first look at the compartmentalised soleus muscle to provide reference values for further modelling.
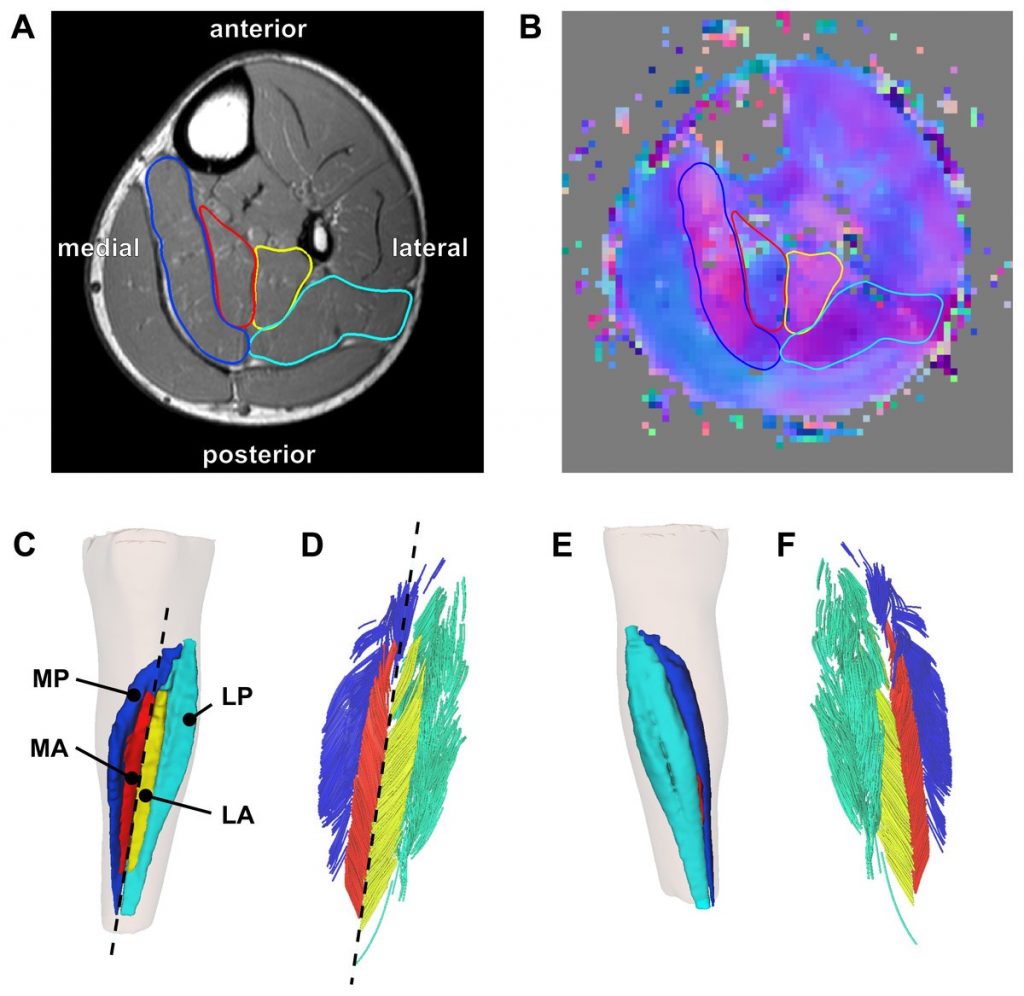
Reconstruction of the architecture of the human soleus muscle using MRI and DTI, taken from ref., showing (A) the MRI slice (midway between ankle and knee) and (B) the corresponding DTI slice taken on a healthy child participant, with (C – F) showing the 3D reconstruction of the surface of all muscle compartments based on the outlines on the anatomical scan.
The human soleus muscle is particularly difficult to study using conventional techniques, such as ultrasound, due to the depth of the anterior and proximal compartments and difficulty in accurate orientation. Hence, DTI is an ideal method to quantify the macroscopic arrangement of muscle fibres of the soleus and help develop comprehensive, quantitative atlases of human muscle architecture.
Prof Herbert’s team have recently used the method to investigate the leg muscles of children with cerebral palsy. Measurements of the medial gastrocnemius muscles were obtained from structural MRI and DTI scans of 20 children with unilateral spastic CP and 20 typically developing children. The study showed that children with unilateral spastic cerebral palsy had reduced range and muscle volume in the calf on the more affected side compared to typically developing children.
The calf plays a vital role in standing and walking, and the differences detected here provide insight into the pathophysiology of muscle contractures and functional impairments in children with cerebral palsy. This knowledge is essential for orthopaedic surgeons and physiotherapists supporting affected children in learning to walk independently.
For further information, please contact NIF Fellow Dr Michael Green.
This story was contributed by NeuRA.
Reviving an ancient Egyptian Mummy sounds like something out of a science fiction movie, but researchers at the University of Melbourne have done the next best thing. In a multidisciplinary project with the Faculty of Medicine, Dentistry and Health Sciences headed by Dr Varsha Pilbrow, the head of Meritamun – a young Egyptian woman who lived more than 2000 years ago – has been imaged using CT and reconstructed using 3D-printing technology.
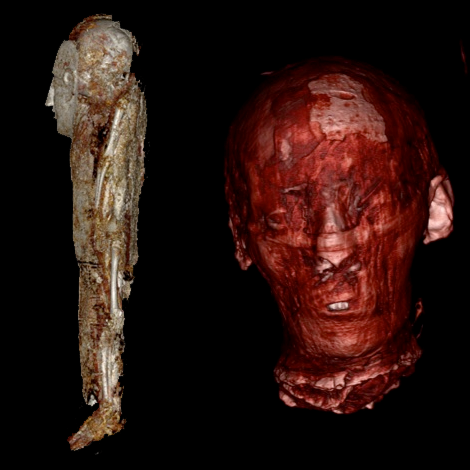
On the left, CT reconstructions of a baby mummy (still being studied). On the right, the CT reconstruction of the head of the mummy named Meritamun from the University of Melbourne’s collection within the School of Biomedical Sciences
Without disturbing the rare specimen and adhering to the controls and procedures of the Human Tissue Act 1982, Meritamun was imaged using the Siemens Human PET/CT in the University of Melbourne National Imaging Facility (NIF) Node. Digital and volumetric displays of tomographic data were acquired and reconstructed for 3D printing to create a skull replica.
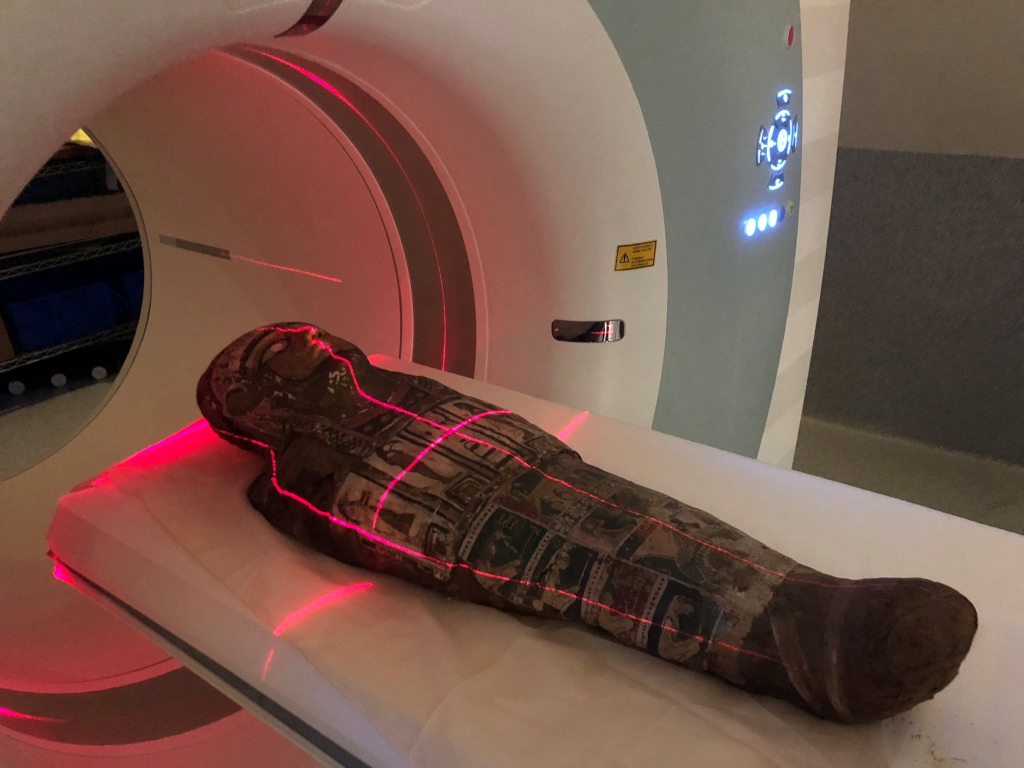
The scanning of a mummy with no adverse affects
Biomedical science Masters Student Stacey Gorski has used the CT data to diagnose Meritanum with anaemia, and hypothesizes a cause of death due to parasitic infection. “The fact that she lived to adulthood suggests that she was infected later in life,” says Gorski. She and supervisor Dr Pilbrow are continuing the investigation, hoping to learn more about the life and death of the ancient Egyptian using forensic pathology.
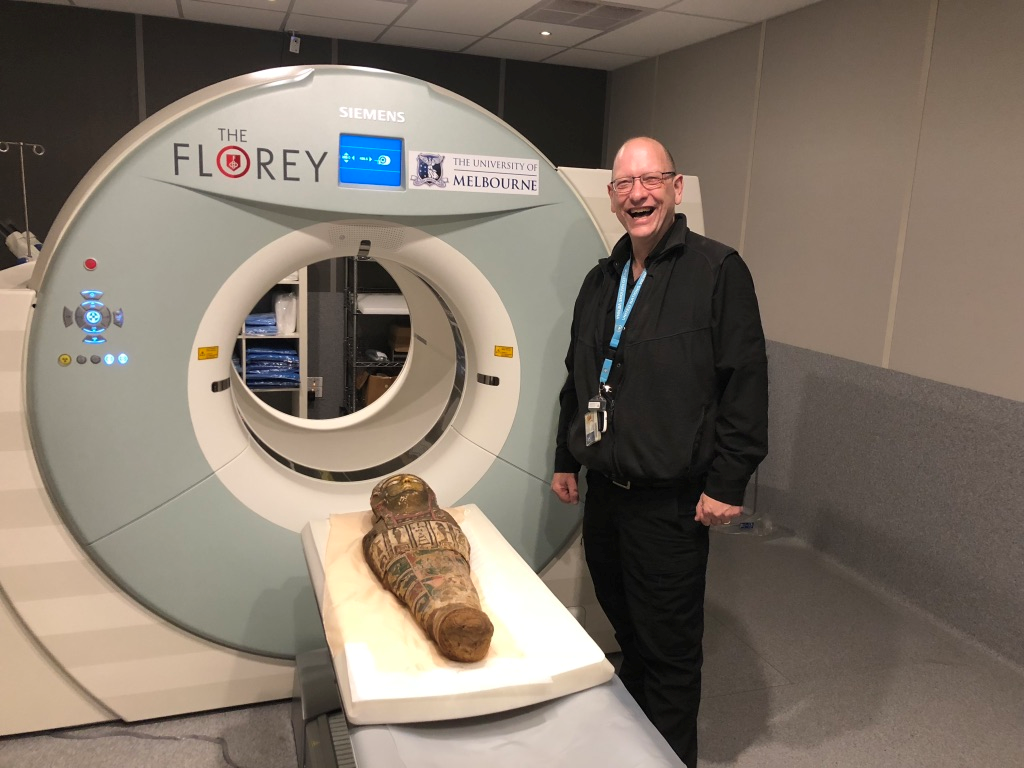
NIF Facility Fellow Mr Rob Williams facilitating access and providing expertise to the Human PET/CT scanner
In addition to learning more about the pathology and environments of population groups of 2000 years ago, the capability to replicate body parts and organs from CT imaging of specimens offers an opportunity for students to interact with old rare samples without damage to the original.
This story was contributed by the University of Melbourne. Acknowledgements go to Varsha Pilbrow, Julietta Capodistrias, Nina Sellars, Quentin Fogg, Michelle Gough, Gavan Mitchell, Peter Mayal, and Natalie Langowski.
For more information, please contact Rob Williams.
Researchers at the Australian Research Council Centre of Excellence for Coral Reef Studies, the University of Western Australia, Curtin University, Aix-Marseille University in France, and the US National Oceanic and Atmospheric Administration wanted to know the history of coral bleaching events on the Great Barrier Reef and how corals are responding to climate change.
Hard long-lived corals, such as Porites, are the backbone of reef ecosystems such as the Great Barrier Reef. Such reef-building corals are sensitive to light levels and temperature. Reef-building corals are already reaching their limits with every heatwave. With ocean temperatures rising, can they hope to survive more frequent extreme temperature events?

Dr. Thomas DeCarlo drilling a 2+ meter core from a massive coral on the Great Barrier Reef
The team of researchers collected Porite core samples across the northern Great Barrier Reef, the Coral Sea, and New Caledonia, bring them to the WA NIF Node at the University of Western Australia, where they were scanned on the Bruker Skyscan 1176 in vivo micro-CT. 3-dimensional image stacks of density variations revealed ‘bands’ within the coral skeletons, corresponding to age. Also seen were high/low density ‘stress bands’, corresponding to environmental stressors such as exceptionally high water temperature.
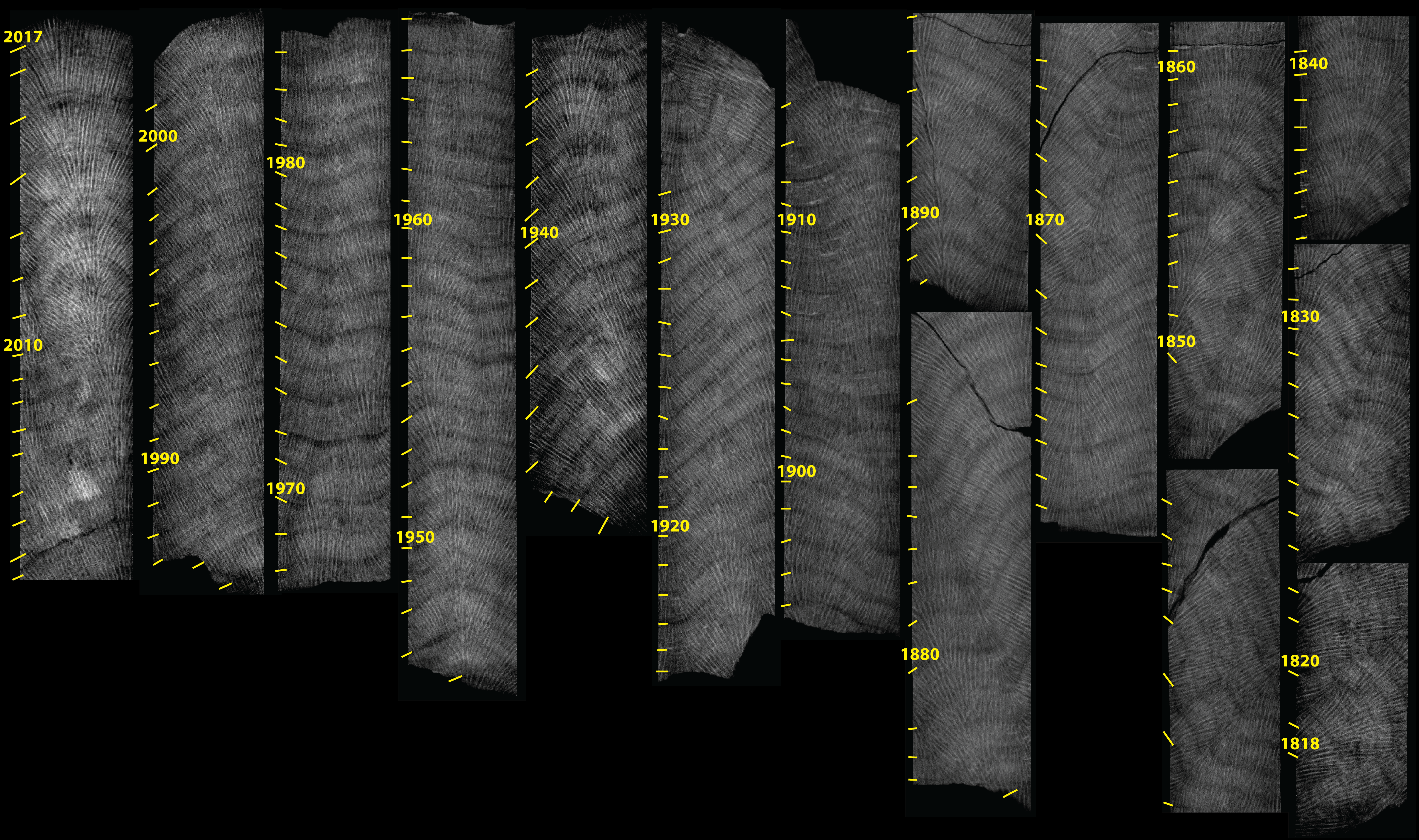
μCT scans (dark/light shading = low/high density) reveal high-density stress bands and partial mortality scars preserved within the skeletons of long-lived Porites corals.
Some of the oldest (and longest) core samples had over 200 annual bands, meaning they were a living coral that has been growing for two centuries. By comparing stress bands to age, change-induced bleaching episodes were mapped, providing a timeline of coral bleaching events. Three striking observations followed: First, the researchers found the first piece of evidence that coral bleaching has occurred prior to the 1980s. Second, a significant increase in the frequency of stress bands was seen over time, consistent with the effects of global warming sparking more frequent coral bleaching events. Finally, recent (within the past few years) acclimatization was seen, whereby corals became less sensitive to heat stress follow repeated exposure to marine heatwaves. These results, published in early 2019, offer hope that reef-building corals surviving heatwaves may be able to adapt for future heatwaves.
This story was contributed by the University of Western Australia. For enquiries, please contact Ms Diana Patalwala.
Sunny Queensland has a high rate of skin cancer, with melanoma as the second most common cancer in Queenslanders. In the area of the head and neck, this can lead to invasion of facial nerves and spread via the base of the skull to reach the brain stem. The extent of the progression, the so-called perineural spread, defines the therapeutic approach, informing the extent of required resection, and the complexity and duration of the surgical procedure. Imaging the extent of nerve involvement is critical to guiding treatment decisions and MRI neurography is the accepted imaging modality of choice.
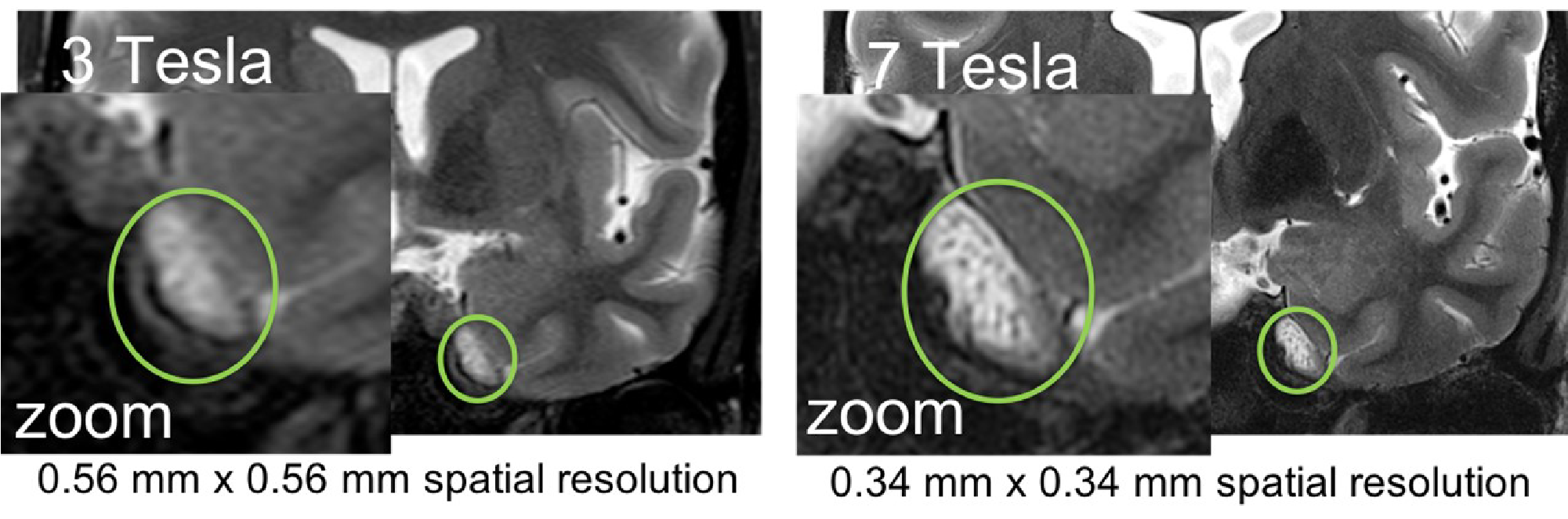
Standard MRI may underestimate disease extent, but a current clinical trial explores the improved resolution and sensitivity using the 7 Tesla ultra-high field MRI scanner at the Centre for Advance Imaging at UQ. This improves visualisation of cranial nerves (see Figure and insert with zoomed nerve fibres) and potentially improves the definition of the perineural spread. The clinical trial is led by Dr. Sommerville from the Royal Brisbane and Women’s Hospital, QLD, and involves a Brisbane-based multidisciplinary team of head and neck surgeons, radiologists and scientists in the UHF MR Research team (Head: Dr. Barth, NIF Fellows Dr. Bollmann and Dr. Ali) at the UQ Centre for Advanced Imaging. The first patient scan has been performed, with 20 patients more to follow after a successful funding application sponsored by the 2018 round of the RBWH Diamond Care Grants.
If you would like to know more about skin cancer, please check the Cancer Council website and speak to your GP.
This story was contributed by the University of Queensland. For further information, contact Dr Tonima Ali.
Magnetic Resonance (MR) opens a window for the non-invasive investigation of MR-observable nuclei, i.e. nuclei with a non-zero spin. Common nuclei in organic substances, like Carbon-12 and Oxygen-16; however, possess a zero-spin consequently rendering Hydrogen-1 (commonly referred as ‘proton‘ due to the single proton in the nucleus) the first and most frequently imaged nuclei.
Despite the much lower signal regime, also the potential of imaging ‘other nuclei’, so-called x-Nuclei MRI was approached in the early years of MRI. In 1985, only a few years after the first proton-based images were published, Sodium-23 was the second nucleus in the human body that was imaged non-invasively using MRI by Hilal and coworkers [1]. In spite of these early beginnings, x-Nuclei imaging in general and Sodium MRI, in particular, did not see the same high-paced progression as proton-based MRI techniques. This downturn was mostly caused by significant SNR limitations resulting from the low in vivo concentration and complicated NMR-signal characteristics of Sodium nuclei.
While SNR issues are more and more overcome through hardware improvements, particularly the recent advent of high field systems, the challenging signal characteristics continue to encourage the development of dedicated acquisition methods. Sodium possesses a 3/2-spin and as such exhibits a fast bi-exponential signal decay. Consequently, Sodium MRI sequences are commonly performed in 3D with k-space sampled via centre-out trajectory schemes resulting in inherent drawbacks for sampling efficiency and SNR. While previous research in Sodium MRI method development focused on the optimisation of sampling trajectories, we present a new acquisition method that tackles Sodium image quality improvements via a sequence timing optimisation.
Recently, we introduced zero-gradient excitation ramped hybrid encoding (zGRF-RHE) Sodium MRI, a timing optimisation that utilises the sequence dead-time delay (time-gap between transmit and receive mode) for gradient pre-ramping. This concept improves encoding time across k-space and in so doing alleviates signal decay effects during the acquisition. It utilises a hybrid sequence encoding approach where the central k-space gap, resulting from gradient activity before receiver onset, is filled with Cartesian single point acquisitions. A sequence diagram of zGRF-RHE is displayed in Figure 1.
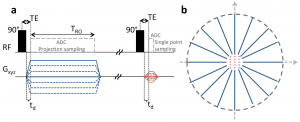
Figure 1: Radial zero‐gradient‐excitation ramped hybrid encoding (zGRF‐RHE) sequence diagram (A) and 2D illustration of hybrid k‐space encoding scheme (B). Gradient‐free excitation is followed by an immediate gradient ramping, data sampling commences after dead‐time. Central k-space is measured separately as single points with the same gradient pre‐ramping condition. TRO, readout duration; td, dead‐time. Figure taken from [2].
Hybrid imaging techniques, like PETRA or its generalised concept RHE, are frequently used in short-T2 proton imaging. Their application to low SNR problems like Sodium was, however, hampered by the employed gradient modulation during signal excitation. In that regard, we establish zero-gradient excitation for an artefact-free excitation profile maximizing image quality. The gradient-free excitation supports high flip angle acquisition, an essential requirement for low SNR imaging like Sodium, but also provides the opportunity to employ advanced RF pulse shapes without introducing a gradient-based excitation bias. Additionally, it should be highlighted that the zGRF-RHE sequence approach essentially describes a timing concept that is not just independent of the RF-excitation but also is not restricted to a particular readout-scheme.
An investigation of 3D-radial zGRF-RHE with standard 3D-radial ultra-short echo time (UTE) imaging can be found in our recent publication [2]. Compared to simulations, phantoms and in-vivo human brain acquisitions confirmed that our proposed sequence timing concept improves on image quality independent of the readout bandwidth and shows reduced image blurring and higher SNR. Figure 2 displays results from an in vivo human brain experiment highlighting improved image sharpness.
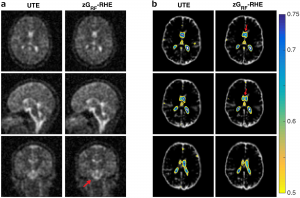
Figure 2: (A) Cross sections through matched slices in in vivo experiments at TRO of 2 ms UTE and zGRF‐RHE. zGRF‐RHE shows sharper details around the brain stem (red arrow). (B) Three consecutive transverse slices of CSF masks with contour lines of normalized UTE and zGRF‐RHE at TRO of 2 ms. zGRF‐RHE provides better delineation between lateral ventricles (red arrow). Figure taken from [2].
In summary, this work introduces an enhanced sequence timing concept with particular applicability to challenging low SNR-problems. Ultimately, this approach is expected to be of use for a wider range of x-Nuclei applications with spin > ½.
For further information, the interested reader is pointed to our full publication which can be accessed at https://onlinelibrary.wiley.com/doi/abs/10.1002/mrm.27484.
References
[1] Hilal, S. K., Maudsley, A. A., Ra, J., Simon, H. E., Roschmann, P., Wittekoek, S., Cho, Z., and Mun, S. (1985). In vivo NMR imaging of sodium-23 in the human head. Journal of Computer Assisted Tomography, 9(1):1–7.
[2] Blunck Y, Moffat BA, Kolbe SC, Ordidge RJ, Cleary JO, Johnston LA. Zero-Gradient-Excitation Ramped Hybrid Encoding (zGRF-RHE) Sodium MRI. Magnetic Resonance in Medicine 2018. https://doi.org/10.1002/mrm.27484.
This story was contributed by Yasmin Blunck of the Melbourne Brain Centre Imaging Unit and Department of Biomedical Engineering, University of Melbourne, Parkville
Copyright 2021 @ National Imaging Facility. All rights reserved.
Read our Privacy Policy.
admin@anif.org.au