Development of Microbeam Radiation Therapy
Only two in ten people diagnosed with brain cancer will survive for at least five years, a figure that has barely improved in the last 30 years. Due to the blood-brain barrier and the risk of damaging normal brain tissue, brain cancer treatments are complex and multidisciplinary. Despite advances in surgical techniques, radiotherapy and chemotherapeutics, brain tumours remain challenging to treat. Hence, image-guided microbeam radiation therapy (MRT) with nanoparticle enhancement is being investigated for the treatment of brain cancer in animal models.
The first long-term pre-clinical study at the Australian Synchrotron
Due to the high collimation and brilliance of synchrotron-generated X-Rays, the beam can be filtered and split into different patterns. MRT is a novel method where the X-ray beam is spatially fractionated by a comb-like collimator into 50 µm X-ray beams spaced 400 µm apart. This creates a “valley and peak” dosage regime that appears more efficacious for killing cancer cells than standard broad-beam therapy. A study conducted by PhD candidate – Elette Engels, at the University of Wollongong [1] examined the anti-tumour effects of microbeam irradiation on the brains of rats inoculated with the 9L glioblastoma cancer, finding a significantly increased survival of the MRT-treated rats compared with untreated rats.
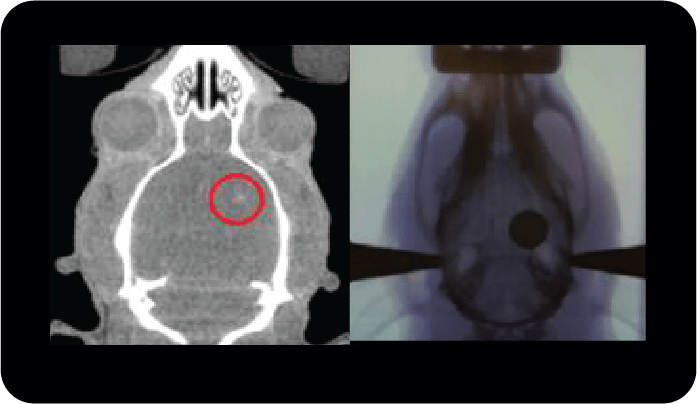
The experiments used ANSTO’s Australian Synchrotron facility (The Australian Synchrotron) to generate the X-Ray beam and the Imaging and Medical Beamline (IMBL) to collimate, quantitate and deliver the MRT dose. NIF Facility Fellows at the NCRIS-enabled Monash NIF node, at Monash Biomedical Imaging (MBI), used the Inveon-CT and the Somotom-CT to image the heads of rats with iodine contrast medium. This established the initial size and location of the tumour burden before MRT therapy. The successful development and verification of an image-guided MRT workflow using instrumentation at MBI and the Synchrotron to deliver an anti-tumour MRT dose accurately to a rat model of glioblastoma serve to establish a baseline for the addition of various radiation enhancing nanoparticles. Further details about the methods developed are available for the advanced reader [1,2].
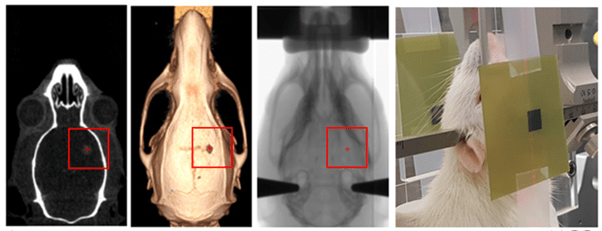
The development of the CT scanning, X-Ray planning and positioning methodology for co-registration of planning images with the microbeam irradiation stage and beam was successful with the confirmation of the registration and image-guided therapy to within 0.3 mm accuracy (Figure 1). Irradiation of rats with intracranial 9L-glioblastoma tumours with MRT produced an average survival of 130 days, compared to the 20-day average survival of the untreated rats (results not shown). The increase in lifespan compared to untreated rats was in excess of 500% and a ‘super rat’ passed away after a long and happy life of more than 300 days. The success of MRT was attributed to successful implementation and development of targeting methodology supplying an adequate tumour coverage with the 15 Gy valley dose.
The successful development of an image-guided MRT workflow linking the radiotherapy infrastructure at IMBL with the imaging infrastructure at MBI has created a unique research environment that has attracted several international studies investigating MRT guided radiation. These include projects with Dr Raphael Serduc from INSERM and the European Synchrotron Radiation Facility, Grenoble, France, Dr Elisabeth Shultke from the University Hospital of Rostock, Germany and Professor Valentin Djonov from the University of Bern, Switzerland. There are also projects closer to home awaiting the lifting of COVID restrictions including a large study with a research group from within ANSTO. While neuronal tumours continue to be very difficult to treat, it is hoped that the promising outcomes from these pre-clinical studies using the unique combination of synchrotron radiation with state-of-the-art imaging will improve future therapeutic outcomes.
This story was contributed by the Monash NIF Node with thanks to NIF Facility Fellows Michael de Veer, Tara Sepehrizadeh and Gang Zheng. We acknowledge the contribution of the IMBL team, in particular Dr Mitzi Klein, veterinarian and IMBL veterinary scientist, and Mr Micah Barnes (IMBL and PeterMac) who developed the image guidance on the IMBL.
For more information on this research, please contact NIF Facility Fellow, Dr Michael de Veer. For more information about brain cancers, please see the Brain Cancer Foundation website or speak with your medical practitioner.
[1] Outputs & Further Reading
- Elette Engels et al., Unravelling Microbeam Radiation Therapy: The first long-term pre-clinical study at the Australian Synchrotron. Conference Presentation at Engineering & Physical Sciences in Medicine Conference (EPSM), Oct 28 – 30, 2019 Perth, Western Australia, Australia.
- Elette Engels et al., Toward personalized synchrotron microbeam radiation therapy. Sci Rep., Jun 1, 2020. doi: 10.1038/s41598-020-65729-z.
- Illawarra News article
- Cosmos magazine article
[2] Methods
The IMBL beamline uses a variable field superconducting magnet arrayto produce a polychromatic x-ray beam from 3 GeV electrons with a beam current of 200 mA. The resulting x-ray beam has an energy spectrum of 20 to 250 keV. Depending on the treatment requirements, the mean energy of the broad spectrum of radiation produced is is tuned by filtering out the lower energy part of the spectrum to keep and use the high energy part. Changing the array peak field from 1.4 to 4T and filtering allows for instantaneous dose rates of 30 – 7,000 Gy s-1.
The IMBL dosimetry was performed using a PTW PinPointTTM ion chamber (model 31015) and a MOSkinTM detector within 255x25x55mm RMI 457 solid water phantoms. The treatment isocentre of the irradiation stage was calibrated using a small steel ball and adjusted until it was centered in all views. The rat is loaded into the MRT field, and a short radiograph is acquired using a Toshiba x-ray tube and Hamamatsu C10900D flat panel detector. The CT scan data identifying the tumour location and size was co-registered with the radiograph of the rat in the MRT field by co-locating skeletal anatomical landmarks. These were the distal tip of the median line, posterior tip of the occipital bone, left and right zygomatic process of the squamosal bone and the left and right anterolateral corner of the frontal bone.
The National Imaging Facility contributed essential instrumentation and imaging expertise to the project. NIFs investments to co-locate imaging hardware at the Monash Node adjacent to the Imaging and Medical Beamline means international and National researchers have access to high-quality imaging hardware and expertise while undertaking studies at ANSTO’s Australian Synchrotron, a partner NCRIS capability.