Understanding Feto-Placental Vasculature
Proper vascular development of the human placenta is crucial for meeting the metabolic needs of the developing fetus during pregnancy. Maternal environmental stressors such as malnutrition disrupt the elaboration of the feto-placental vasculature that, in turn, impacts on placental function and results in reduced fetal growth. The ramifications of this are not only on short-term foetal health but also on long-term health outcomes. Indeed, distortion in placental shape and size strongly associate with later adult health outcomes such as cardiovascular disease, obesity and cancer.
Dr Caitlin Wyrwoll of the School of Anatomy, Physiology and Human Biology, at The University of Western Australia, is leading a multidisciplinary team that is investigating, in rodent models, how everyday environmental stressors in pregnancy alter feto-placental vascular morphology and placental function. Ultimately the team will seek to identify potential therapeutic targets to enhance placental vascular development and then apply this to experimental models to assess the outcomes on fetal development and adult health.
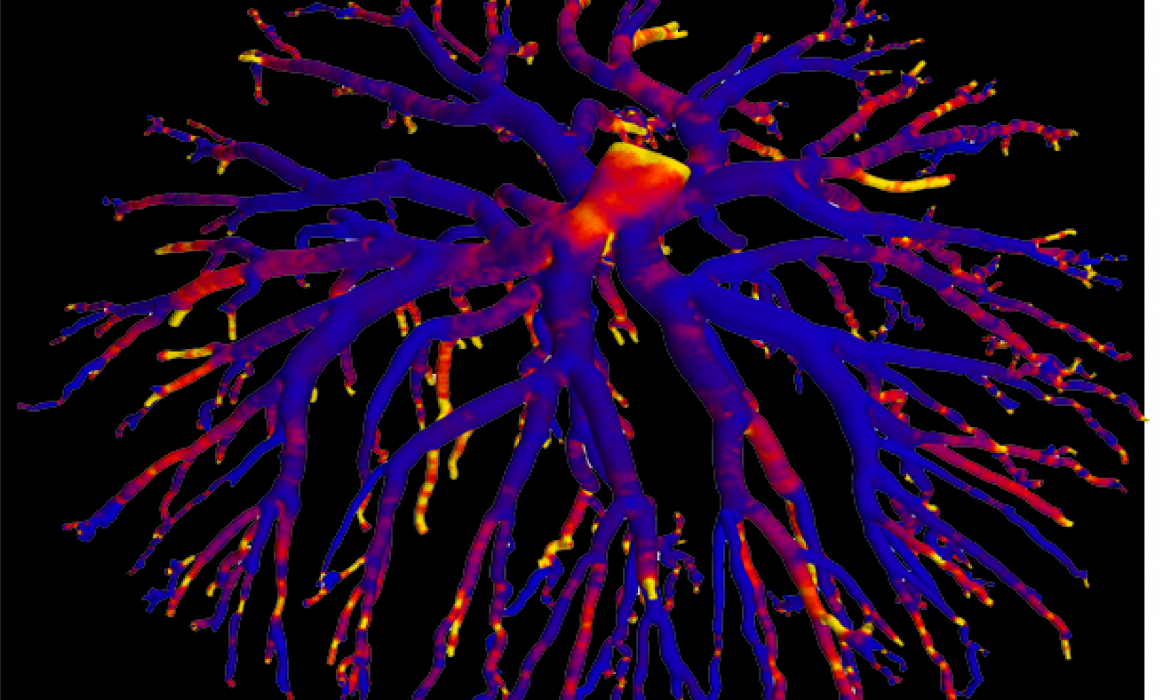
The research project involves collaboration with the Western Australian NCRIS facilities of the National Imaging Facility and Microscopy Australia to image, visualise and characterise the geometry of the arterial and venous feto-placental vascular trees using high-resolution X-ray microscopy (ZEISS Xradia 520 Versa).
Dexamethasone administration during rodent pregnancy is used as a model to simulate excess placental and foetal glucocorticoid exposure (a known effect of prolonged stress). Control and treated animal models are anaesthetised at day 22 of gestation and their uteri collected. The feto-placental units are dissected and foetal anaesthesia induced. The individual feto-placental vascular trees are cleared of blood and perfused with Microfil®, a radio-opaque polymer casting compound. Each cast is stabilised in PBS in a plastic vial and imaged using a wide field-of-view of ~13.4 mm, a voltage of 50kV, more than 3000 projections through 360 degrees, and an exposure time of 7s. The ZEISS XMReconstructor software is used to reconstruct an image volume (standard parallel beam back-projection algorithm) with voxels of size ~7.0 μm.
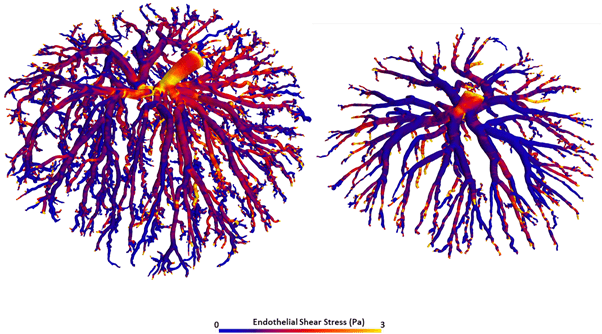
The team have completed a preliminary study involving control and dexamethasone-treated rats and both the venous and arterial feto-placental vasculature trees. A visual comparison of treatment to control indicates that for both types of the vascular tree, there is reduced branching in the fine vessels and reduced vessel density. A quantitative comparison indicates reduced total vessel length and total vessel volume.
A methodology is currently being developed for a more comprehensive and automatic quantitative assessment of vasculature morphology and geometry. This includes automatic segmentation, filtering, centre-line extraction and characterisation of the vessel tree in terms of its branching characteristics such as its branching hierarchy and angles, vessel diameters and the tortuosities of vessel segments. Furthermore, these vascular tree images are being used in a world-first study to model placental blood flow using computational fluid dynamics.
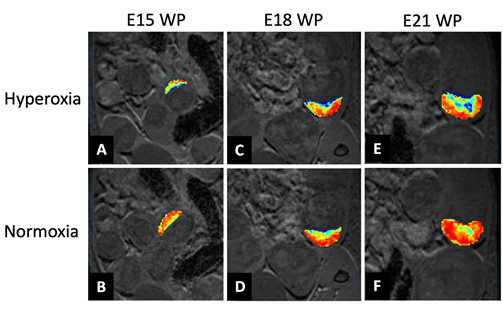
3D vascular casts have been prepared of foetal blood vessels in control and glucocorticoid exposed pregnancy models at three pregnancy time points (E16.5, 18.5 and 21.5). Recent work has revealed that blood flow forces coming from the placenta affect the development of foetal liver, heart and brain structure; particularly of the blood vessels. Using the new high-resolution preclinical ultrasounf at the CMCA, the next step will be to conduct assessments of the placental vessels and foetal heart in control and dexamethasone-exposed pregnancies. The aim is to understand the observed changes more fully as they have important implications for fetal organ function.
For more information on this work, contact Dr Caitlin Wyrwoll or Dr Ivan Lozic/Ms Diana Patalwala.
Related publication: https://www.sciencedirect.com/science/article/pii/S0169260719303293?via%3Dihub
This story was supplied by the UWA NIF Node, in collaboration with: School of Anatomy, Physiology and Human Biology, the University of Western Australia School of Mechanical and Chemical Engineering, the University of Western Australia Centre for Microscopy, Characterisation and Analysis, the University of Western Australia